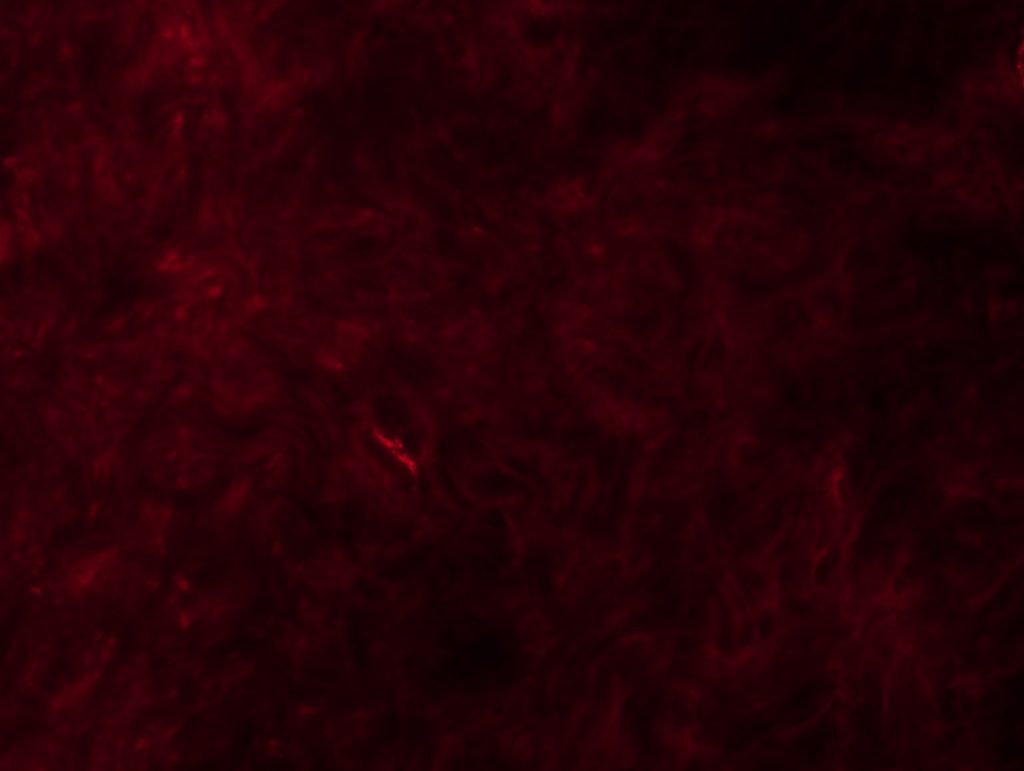
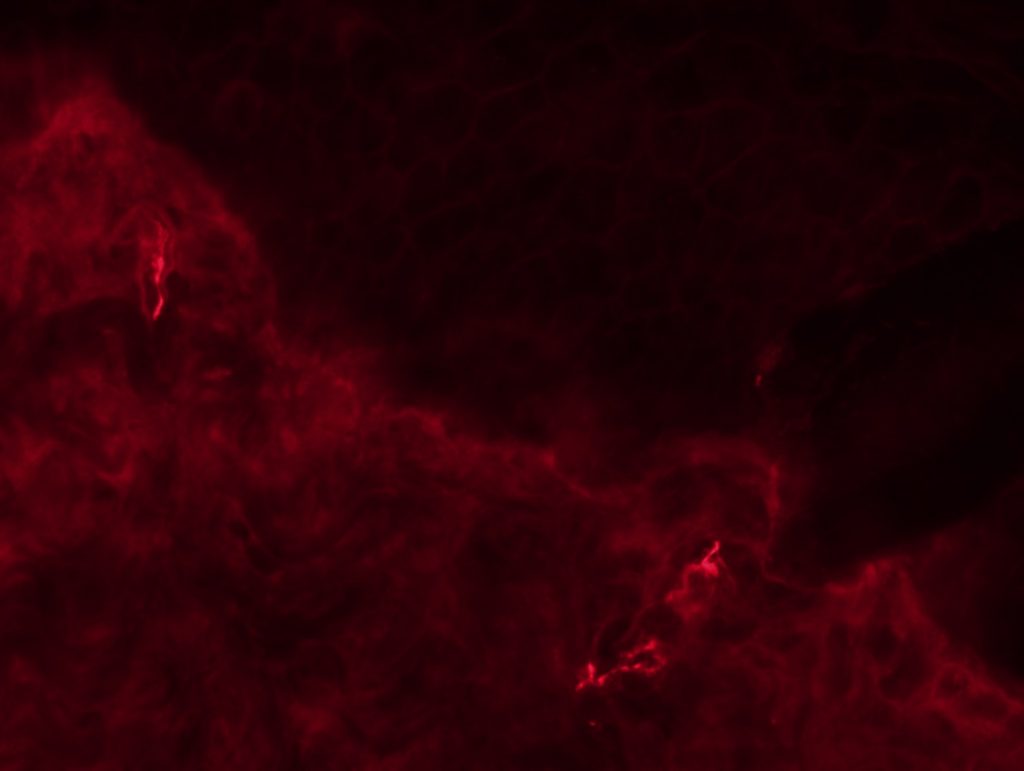
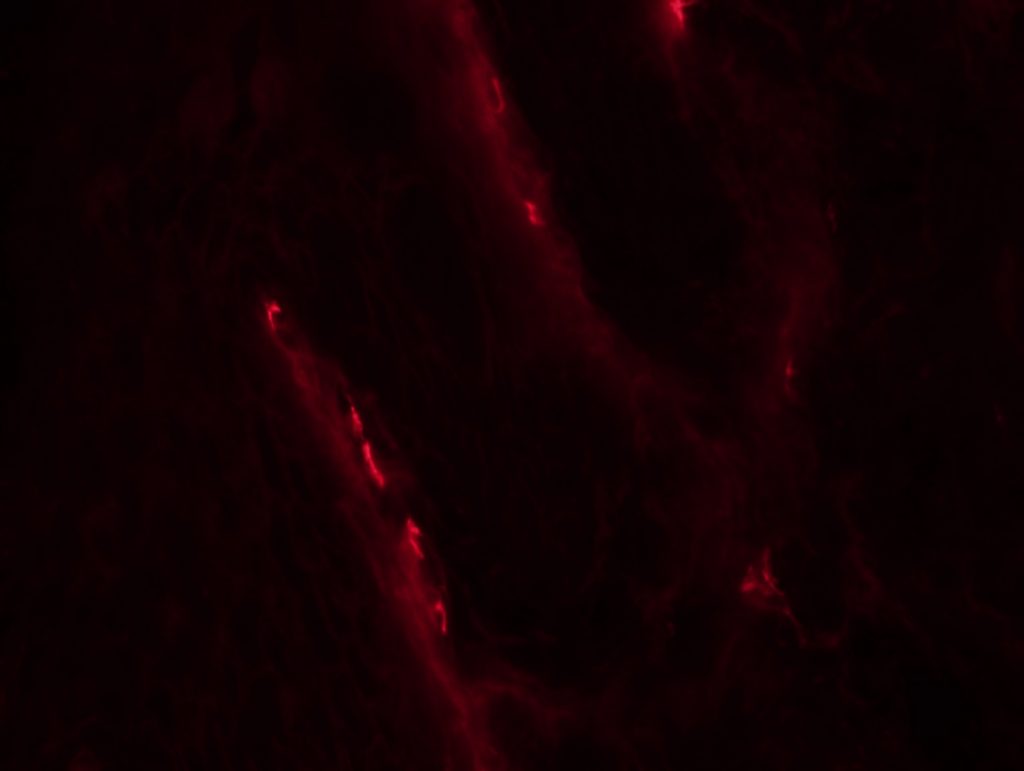
OBJECTIVE: To test myelinated fibers IHC and nodo-paranodes in foot pads from a rat. We will perform 3 different conditions: MBP + caspr, MBP + neurofascin and Pan-Na channel.
IMMUNOFLUORESCENCE (in humid chamber):
1st day:
- Air-dry slides for 30 min and thaw one or two blocking aliquots.
- Outline the sample boundaries with DAKO-PEN.
- Block 30min at room temperature with 10% BSA in PBS.
- Preparation of the primary antibody
a. 1%BSA (1ml 10%BSA+9ml PBS)
b. 1%BSA + 0.3%TritonX (1ml 1%BSA + 3ul TritonX) (vortex).
c. For Caspr + MBP: Caspr1 ab133634 (1:50) diluted in 1%BSA + 0.3% TritonX. MBP (polyclonal mouse-to-human) (1:200) diluted in 1%BSA + 0.3%TritonX. d. For neurofascin + MBP: NF AF3235 (1:100) + MBP (polyclonal mouse-to-human) (1:200) diluted in 1%BSA + 0.3%TritonX. e. For Pan-Na channel S8809 1:100 diluted in 1%BSA + 0.3%TritonX - Remove the blocking solution (tapping on paper, no washing).
- Put the primary antibody on the sections (approx. 40uL in each).
- Incubate at room temperature overnight.
2nd day:
- Wash the slides in PBS (x3) for 5 min.
- Preparation of secondary antibodies – Dilutions in 1% BSA in PBS:
a. Caspr1 + MBP – GAR488 1:100 + GAM594 1:200
b. Neurofascin + MBP: GAC488 1:100 + GAM594 A11005 1:200
c. Pan-Na channel: GAM594 A11005 1:100
- Put the secondary antibody on the slide (40uL) for 2h at room temperature and in the dark.
- Wash with PBS x3.
- Cover the slides with Mounting media: Vectashield-DAPI.
- Cover the edges with nail polish or Vitro-Cloud (to prevent the preparations from drying out).
- Store at 4ºC in the dark until reading. Once analyzed, they are frozen at -20ºC, where they can last for years and can be re-analyzed in the future if necessary.
RESULTS: We can see some nodes/paranodes, although not as clear as I saw them in Wurzburg.