2018.06.04: Coating and seeding
- Prepare 4 plates of 35 mm of diameter with 5 coverslips (12 mm)
- Treatment with:
- Poly-L-Lysine 1h (0’1 mg/ml diluted in Borate Buffer) + Laminin 1h (2’4 microg/ml diluted in Borate Buffer)
- Laminin 1h (2’4 microg/ml diluted in Borate Buffer)
- Poly-L-Lysine 1h (0’1 mg/ml diluted in Borate Buffer) + Laminin 1h (10 microg/ml diluted in Borate Buffer)
- Laminin 1h (10 microg/ml diluted in Borate Buffer)
- Wash with PBS 1x (3 times)
- Seed 15000 cells/cm2 –> every plate has 8 cm2 –> 120000 cells/plate 35 mm (in proliferation medium)
- Incubate at 37ºC for 24h
2018.06.08: treatment and medium exchange
The medium has not been changed before because there were few cells..
Change the medium of the cells –> Add retinoic acid to the differentiation medium (1 microl RA in 1 ml differentiation medium)
*Change the half of the differentiation medium (+ RA) every 2 or 3 days.
2018.06.11: treatment and medium exchange
Change the medium of the cells –> Add retinoic acid to the differentiation medium (1 microl RA in 1 ml differentiation medium)
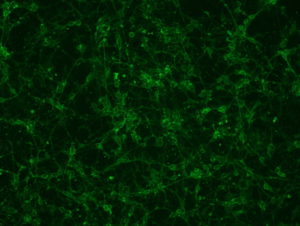
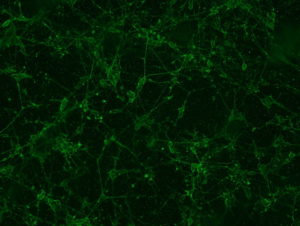
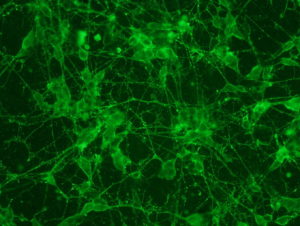
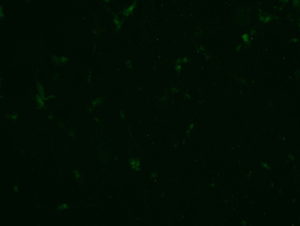
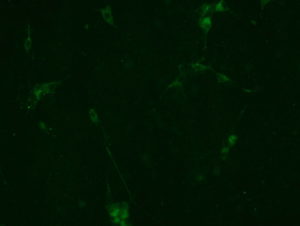
2018.06.13: ICC
- Take 2 coverslips of each condition and put them in a 24 well plate
- Fix with PFA 4% (aprox 1 ml/well). Incubation at RT during 20 min.
- Wash with PBS 1x
- Patient’s sera diluted 1/100 with differentiation medium (positive and negative control)
- Wash with PBS 1x
- Secondary antibody: GAH 488 (IgG or IgM) diluted 1/1000 in Goat Serum 5 %
- Wash with PBS 1x
- Vectashield mounting medium
Result: the cells are well preserved in all the conditions (except Poly-L-Lysine + Laminin 10 microg/ml ). From now on, we will use the laminin 2’4 microg/ml coating.